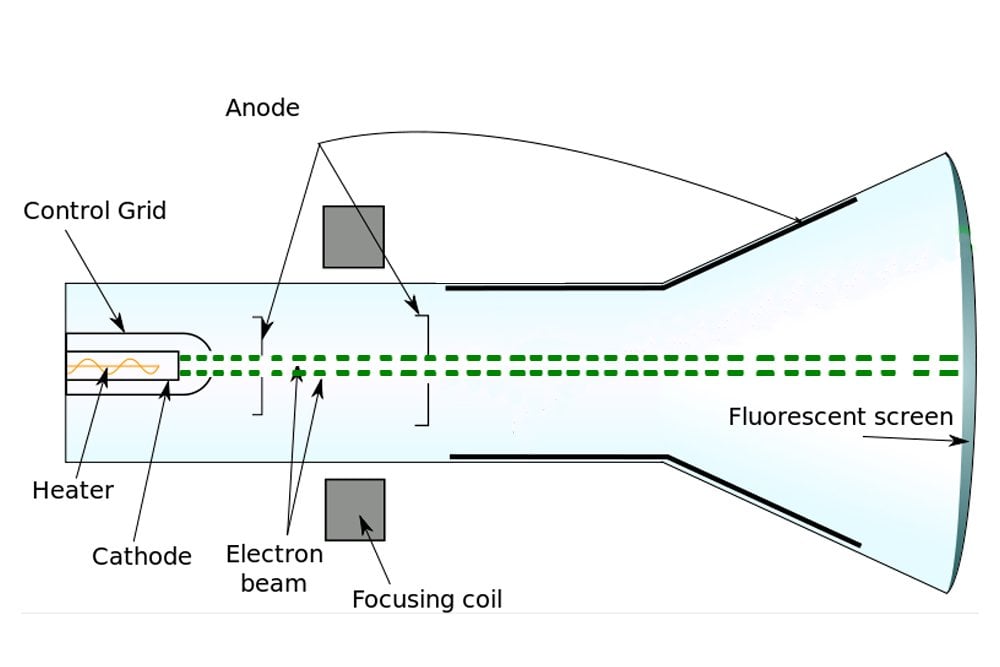
He left research in 1918 to become Master of Trinity College. When the cathode rays are deflected by the electrostatic field, the phosphorescent band breaks up into several bright bands separated by comparatively dark. In addition to being awarded the Nobel Prize in 1906, he was knighted in 1908 by King Edward VII. Thomson published 13 books and more than 200 papers in his lifetime. They had one daughter, Joan, and one son, George Paget Thomson, who went on to become a physicist and win a Nobel Prize of his own. Thomson detected their path by the fluorescence on a squared screen in the jar. Cathode rays were produced in the side tube on the left of the apparatus and passed through the anode into the main bell jar, where they were deflected by a magnet. Thomson married Rose Paget, one of his students, in 1890. Thomson first investigated the magnetic deflection of cathode rays. This was the first use of mass spectrometry. In doing so, he discovered that neon was composed of two different kinds of atoms, and proved the existence of isotopes in a stable element.

This led to one of his other famous discoveries in 1912 when he channeled a stream of ionized neon through a magnetic and an electric field and used deflection techniques to measure the charge to mass ratio. In 1906, Thomson began studying positively charged ions, or positive rays.

